|
1/27/2024 0 Comments Negative electron affinity
The electron affinity of the iodine atom is not much different from those of the. 
Thus, metallic character increases as we move down a group and decreases across a period in the same trend observed for atomic size because it is easier to remove an electron that is farther away from the nucleus. If Negative: no color change-no starch in the food being tested. Most adsorption sites give a negative electron affinity (NEA), with the largest NEAs 1.47 eV on the bare surface (1 ML coverage) and 1.36 eV on the. A fluorine atom in the gas phase, for example, gives off energy when it gains an electron to form a fluoride ion. 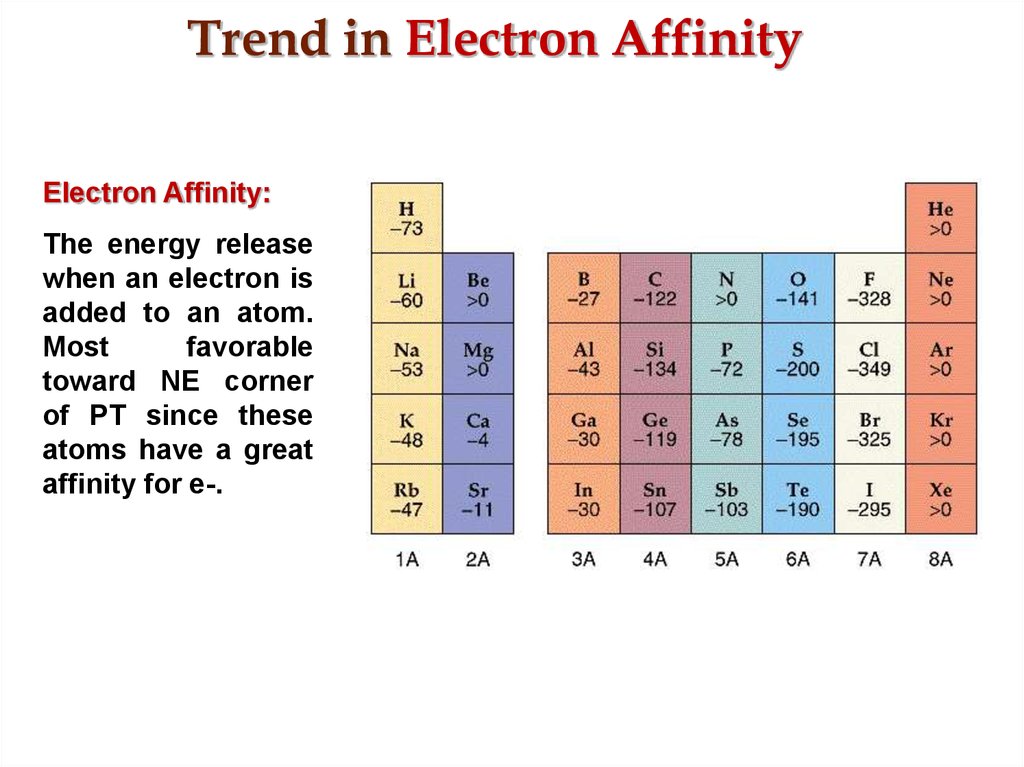
Metallic properties including conductivity and malleability (the ability to be formed into sheets) depend on having electrons that can be removed easily. The electron affinity of an element is the energy given off when a neutral atom in the gas phase gains an extra electron to form a negatively charged ion. The gray squares represent metals, the orange squares are "metalloids" and the yellow squares are nonmetals.Ĭredit: "Various Periodic Trends" by Sandbh is licensed under CC BY-SA 4.0 \): Metallic character increases as you go from right to left on periods and move down groups on the periodic table. Some atoms readily attract electrons, and the electron affinity has a negative value, meaning that energy is released.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
